The European MedTech sector has around 27.000 Med-Tech companies, 95% of which are SMEs. These companies need to demonstrate not only safety and performance over the entire life cycle of a product, but also, tangible benefits for the patients and the healthcare system. The new Medical Device Regulation (MDR) increases as well the requirements related to the reliability in relation to its intended clinical use placing a heavy burden on medical device innovation in Europe.
Being the MD’s sector a highly innovative one, with an average lifecycle per product of 18-24 months, this scenario represents several challenges, especially for high-tech SMEs.
SMEs are particularly affected by MDR’s extensive documentation and report obligations and new clinical testing requirements. With the increased demands and their associated complexity, a significant number of SMEs in the MedTech field are now economically and resource wise at risk, which threatens value chains and thus has the potential to affect the sustainability of the entire European medical device sector. There is potential for a massive loss of innovation, jobs, economic strength and product diversity.
CIDETEC is participating in 2 European Project that aim to minimize the impact of the MDR to SMEs, while maintaining a high level of safety for the patients and robustness for the processes.
These 2 projects are “Open Innovation Test Beds” meaning a network of services that covers a part or the total of the development value chain, and help companies to arrive to market at an accelerated pace.
TBMED
TBMED is a 4-year-long project coordinated by CIDETEC Nanomedicina that has received funding from the European Union’s Horizon 2020 research and innovation programme. Its aim is to increase the access of high-risk medical devices to patients, by helping SMEs to minimize the market approval and reimbursement process time, optimizing the process of transforming a prototype (TRL4) into a valuable innovative MD (TRL7).
TBMED will establish an Open Innovation Test Bed (OITB) consisting of a connected network of labs providing a single entry point to services along the whole value chain, from preclinical development to clinical testing, based on Quality-by-Design (QbD).
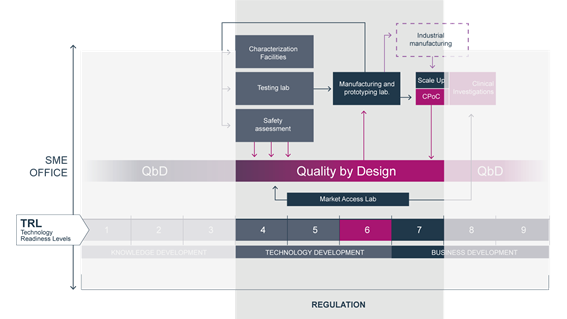
The objectives of the service platform are:
- Increase the quality and reduce the risk of MDs and facilitate subsequent clinical testing
- Build the arguments to demonstrate real benefits (value / final outcomes) of the new devices to increase their success in entering the market
- Reduce cost and variability of the manufacturing process and the speed of product release to the market by carrying out statistically DoE for process validation.
In order to develop the platform, 3 use cases in different TRL stages are used:
- Keratoprosthesis: a biocompatible and flexible synthetic cornea (TRL 3)
- Glycobone: an injectable ready-to-use hydrogel tailored for oral bone defect reconstruction to be developed for 2 different indications (TRL 4/6)
- Hyperthermia treatment for colorectal cancer, based on magnetic nanoparticles and alternating magnetic field (TRL 3/4 )
The OITB will be developed during the course of the project and will become a commercially available platform after the project is finished providing services in areas such as technology development, business support and financing.
A consortium consisting of 5 Research groups (CIDETEC, University of Zaragoza, INSERM, CURAM, FRAUNHOFER), 5 SMEs (ANTARES, SILAB, CYBERNANO, nB, EURICE), 1 industry (AJL) and 2 clinical research organizations (BIOEF, ECRIN), all of them with significant track record in knowledge creation and innovation in their respective domains, guarantee the successful implementation of this project and the creation of the OITB as a separate legal entity afterwards.

Website: www.tbmed.eu
MDOT
MDOT is a 5 years collaborative project in the framework of DT-NMBP-02-2018 Open Innovation Test Beds for Safety Testing of Medical Technologies for Health (IA). The goal of MDOT is to implement a platform as a meta-network to preserve Med Tech innovation and economic strength, reduce animal testing, and support MDR´s new level of patient safety.
The goal of MDOT is to establish a ‘one-stop-shop’ platform that enables MedTech SMEs to:
1) better know the regulatory
requirements for safety assessment of their medical devices
2) establish a database of detailed testing data of commonly used materials and
components
3) gain access to advanced testing and fabrication methods
4) perform evaluations of commonly used parts together, and
5) safely and transparently exchange testing data using state-of-the-art
technology to decrease cost while increasing quality and safety of medical
device innovations.
Operability of MDOT will be demonstrated using three demonstrators:
1) A test bed for the unmet
medical need of aerosol delivery through inhalers specifically designed and
adopted for infants, term and preterm neonates to allow tests under
standardised conditions, which does not yet exist.
2) A test bed for the long term accelerated testing of polymeric active
medical devices inaccessible with current technologies due to the temperature
sensitivity of polymers that will be also transformed into prediction software
modules.
3) A test bed for increasing the safety and lifetime of total joint
arthroplasty that involves state of the art testing protocols on wear
measurement and computational modelling.
Partnership:
The MDOT platform design and development, profits greatly from the specialised expertise present within the MDOT consortium. Together, the partners cover the value chain from basic and applied research and testing (ITEM, CIDETEC Nanomedicine, University of Bayreuth, University of Linz, University of Leeds, University of Southampton), to MedTech industry (DEMCON, ACMIT, MATHYS), regulatory affairs experts (MT-Consult, Nanoconsult), and clinical application (University of Poznan, Hannover Medical School).

Our role:
– Developer of one of the demonstrators: Hydrophilic coating to control wear of the prosthesis.
– Head of the Orthopaedic Node inside the testing bed: Communication and MDR.
Website: https://www.item.fraunhofer.de/en/press-and-media/press-releases/pm-mdot.html


