The partners of RESTORE are active also in other international projects on nanomedicines and nanoenabled medical devices. Very recently, the SINTEF partner co-authored a publication from the REFINE project (http://refine-nanomed.eu/), describing methodological needs for characterization of nanomedicines and nano-enabled medical devices. The latter is directly related to the systems used in RESTORE, which are macroscopic implants functionalized with nanomaterials.
Suitable standardised methods need to be available for quality and safety assessments of these innovative products prior to their translation into the clinic and for monitoring their performance when manufacturing processes are changed. The question arises which technological solutions are currently available within the scientific community to support the requested characterisation of nanotechnology-based products, and which methodological developments should be prioritized to support product developers in their regulatory assessment.
First, only standardised methods were considered, leading to the identification of methodological needs in five areas of characterisation, including: (i) surface properties, (ii) drug loading and release, (iii) kinetic properties in complex biological media, (iv) ADME (absorption, distribution, metabolism and excretion) parameters and (v) interaction with blood and the immune system. In the second step, a detailed gap analysis included analytical approaches in earlier stages of development, and standardised test methods from outside of the nanotechnology field that could address the identified areas of gaps. Based on this analysis, three categories of methodological needs were identified, including (i) method optimisation/adaptation to nanotechnological platforms, (ii) method validation/standardisation and (iii) method development for those areas where no technological solutions currently exist.
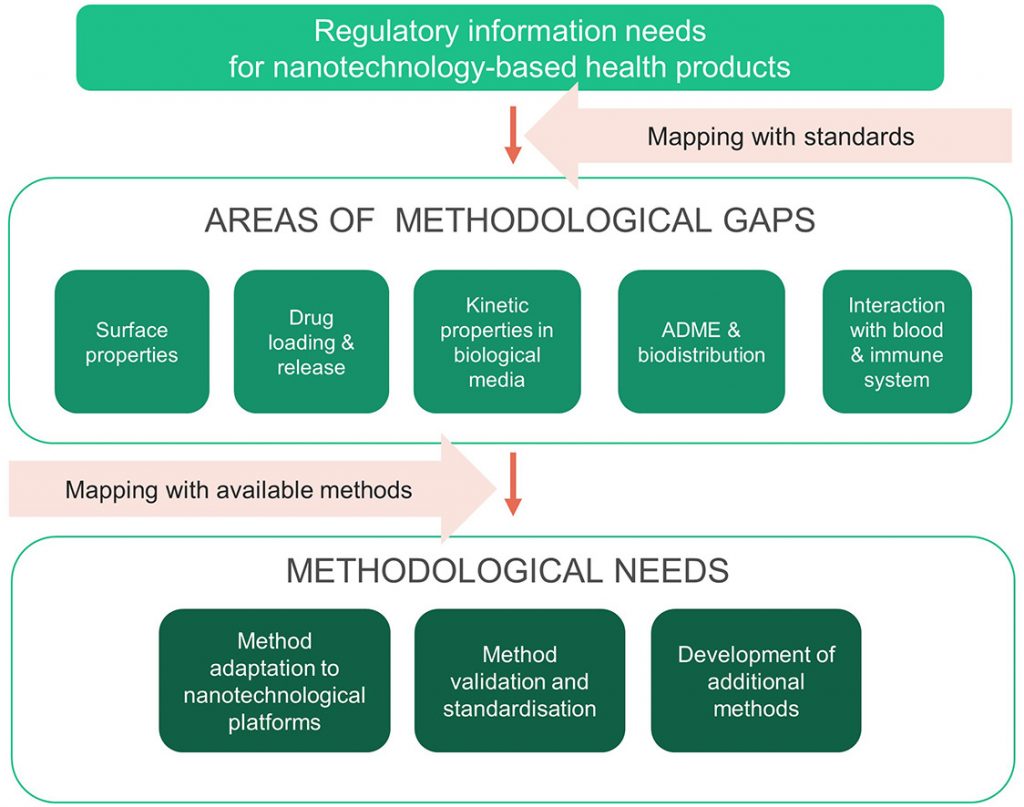
The work also identified a need for coordinated, international efforts towards formal standardization of methods, but does acknowledge that this is a challenging task, particularly for biological tests like immunological assays.
Original paper: B. Halamoda-Kenzaoui et al.; Journal of Controlled Release (2021), 336, pp. 192–206.https://doi.org/10.1016/j.jconrel.2021.06.016


